8 Swimming Levels To Learn for Lifelong
Whether you’re a child, teen, adult, or senior, learning to swim is an important life skill. Knowing how to swim not only can save your life but also the lives…
5 Best Ginger Juicer For Your Health: Top Picks & Reviews
Ginger is a largely popular ingredient in every kitchen. Let’s find out Best Ginger Juicer for Your Health. From cooking to health issues, ginger has loaded with A-graded benefits. The…
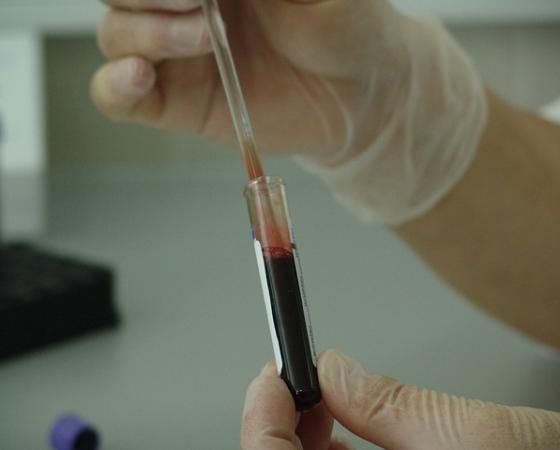
8 Types of Cancer Treatments
8 Types of Cancer Treatments currently used in medical world. Get knowledge on Types of Cancer Treatments, care for your body, don’t overuse it, be smart, eat smart. Daily news…
Your Guide To Cruise Travel: Everything You Need To Know
In our previous article, Breathtaking Images of Travel, we listed several destinations worldwide, from the Blue Domes of Oia in Santorini to Murcielago Island in Costa Rica. These destinations are…
Which is The Best Hotel Of World | Lists of 50 Best Hotels of the World
The “Best Hotel of World” turned into be built by a pope and was regularly visited with the aid of one of the most popular, Italy’s very well-known opera composers.…
Multilayer PCBs: Unveiling The Technological Marvel Behind Modern Electronics
The demand for smaller, faster, and more powerful devices continues to grow in the fast-paced world of modern electronics. This technological breakthrough centres around the Multilayer PCB (Printed Circuit Board).…
Why Camcorders Are Making a Comeback in the Digital Age
In today’s digital age, it seems that everyone has a video camera in their pocket with the prevalence of smartphones. However, there has been a notable resurgence in the popularity…
Food Fighting Cancer Cells | How Does Cancer Develop
Food fighting cancer cells are Apples, Berries, Cruciferous vegetables, Carrots, Fatty fish, Walnuts, Legumes, Supplements, and medications. Cancer is a condition where certain cells in the body grow uncontrollably and…